 During BCR signaling, the phosphorylation by SYK at Y551 enhances the catalytic activity of BTK and initiates subsequent Y223 autophosphorylation. BTK has two critical tyrosine phosphorylation sites, Y223 in the SH3 domain and Y551 in the kinase domain. BTK comprises five different protein interaction domains, which include an amino-terminal pleckstrin homology (PH) domain, a proline-rich TEC homology (TH) domain, SRC homology (SH) domains SH2 and SH3, and a kinase domain (Fig. The process of B cell development and the role of BCR in this process are described in Fig. BTK loss-of-function mutations result in X-linked agammaglobulinemia, a type of immunodeficiency that is characterized by the lack of mature B cells and immunoglobulins and consequent opportunistic infections in young boys, highlighting the importance of BTK in B cell development and humoral immunity. BTK is a member of the Tec family kinases, which contain interleukin-2-inducible T cell kinase (ITK), tyrosine kinase expressed in hepatocellular carcinoma (TEC), resting lymphocyte kinase (RLK), and bone marrow expressed kinase (BMX). BTK was originally identified as a non-receptor protein tyrosine kinase in 1993. The essential role of BTK in BCR signaling makes it an ideal target for suppressing BCR signaling. 
In this review, we discussed the rationale for the use of non-covalent BTK inhibitors and the preclinical and clinical studies of non-covalent BTK inhibitors in B cell malignancies. Preliminary clinical studies have suggested that non-covalent BTK inhibitors are effective and well-tolerated. Non-covalent BTK inhibitors do not bind to C481, therefore providing a potentially effective option to patients with B cell malignancies, including those who have developed resistance to covalent BTK inhibitors. Alternative therapeutic options are needed if acquired resistance or intolerable adverse events occur. Additionally, adverse events due to the off-target inhibition of kinases other than BTK by covalent inhibitors are common. A major mechanism for the acquired resistance is the emergence of BTK cysteine 481 (C481) mutations, which disrupt the binding of covalent BTK inhibitors. However, acquired resistance to covalent BTK inhibitors is not rare in B cell malignancies. Several covalent BTK inhibitors have shown remarkable efficacy in the treatment of B cell malignancies, especially chronic lymphocytic leukemia. Bruton tyrosine kinase (BTK) is a key component of BCR signaling, establishing BTK as an important therapeutic target. 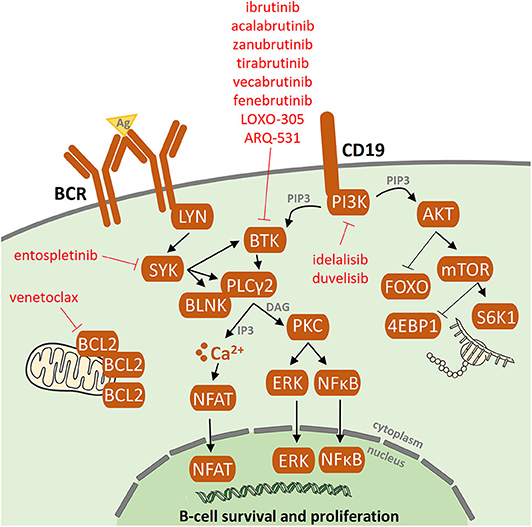
Activation of BCR signaling promotes the survival and proliferation of malignant B cells. B cell receptor (BCR) signaling is involved in the pathogenesis of B cell malignancies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
